PrecisionMRX® Nanoparticles
Superparamagnetic iron oxide nanoparticles that are, quite simply, super.
How it Works
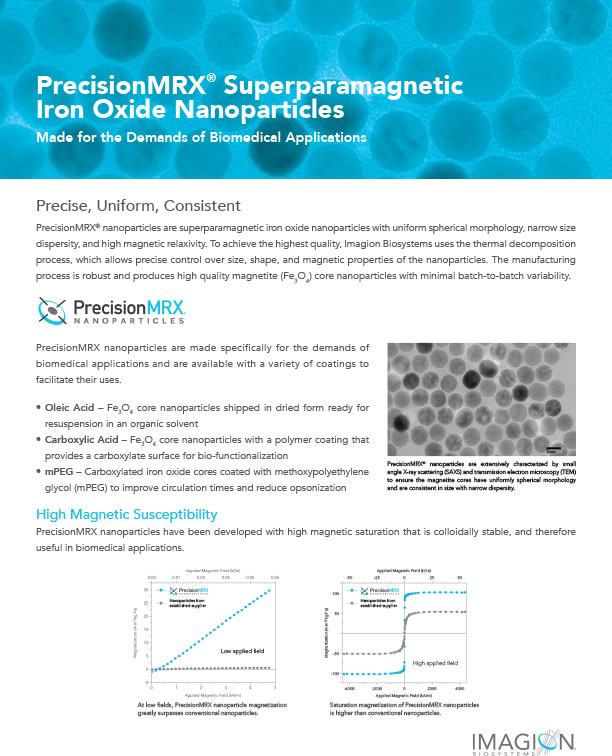
PrecisionMRX® nanoparticles are extensively characterized by small angle X-ray scattering (SAXS) and transmission electron microscopy (TEM) to ensure the nanoparticle cores have a uniformly spherical morphology and are consistent in size with narrow dispersity, resulting in minimum batch-to-batch variability.
Particles from other suppliers typically have poor control over size and shape making them less reliable and consistent for the demands of many applications.


Conventional
Suppliers

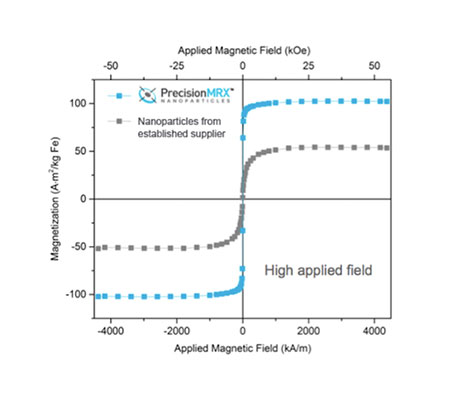
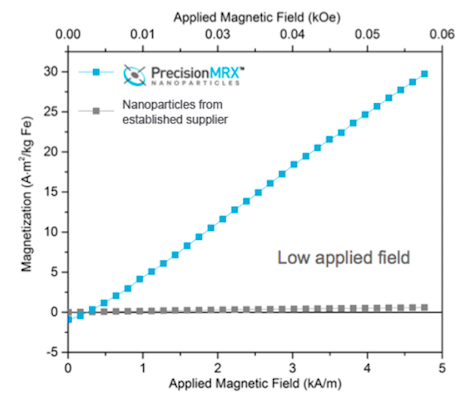
High Magnetic Susceptibility
PrecisionMRX® nanoparticles have been developed for magnetic relaxometry and other demanding biomedical applications where the magnet susceptibility of the nanoparticle can be instrumental.
The saturation magnetization of PrecisionMRX® nanoparticles is typically twice as great as conventional iron oxide particles, even in an applied low or weak magnetic field.
PrecisionMRX® nanoparticles can provide excellent contrast even at 1.5T.
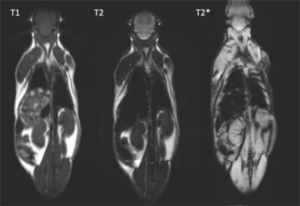
By using different MRI sequences, clear information on anatomy (T1-weighted), nanoparticle location (T2-weighted) and magnetic susceptibility (T2*) can be achieved.
Lot-to-Lot Consistency
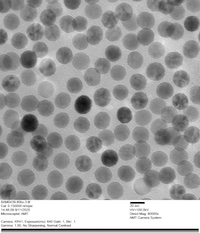

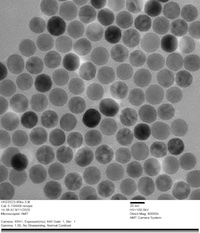

SPIONS are ideal for various biomedical applications
Explore the many applications of our PrecisionMRX® nanoparticles and find answers to common questions in our FAQs.
Featured Products
-
$369.00 – $660.00 Select options This product has multiple variants. The options may be chosen on the product page
-
$369.00 – $689.00 Select options This product has multiple variants. The options may be chosen on the product page
-
$450.00 – $1,679.00 Select options This product has multiple variants. The options may be chosen on the product page
-
$369.00 – $660.00 Select options This product has multiple variants. The options may be chosen on the product page

Transforming medical imaging for early disease detection
We’re on a mission to make cancer more detectable.