Imagion Biosystems Hits Major Milestone, Submits IND Application to U.S. FDA for Phase 2 Trial of MagSense® HER2 Imaging Agent
Key Highlights:
- Imagion Biosystems has lodged its Investigational New Drug (IND) application for the MagSense® HER2 Imaging Agent to the U.S. Food and Drug Administration (FDA)
- IND Submission marks a major milestone in the clinical development of the company’s proprietary MagSense® platform
- Phase 2 Clinical Trial enrollment planned to commence following FDA approval
- Strategic trial partners engaged, study site selection process has begun
Imagion Biosystems (ASX: IBX) (Company or Imagion), is dedicated to improving healthcare outcomes through early and more accurate detection of cancers utilising its proprietary MagSense® imaging technology. The Board is pleased to provide shareholders with an important update on its progress towards commencing its MagSense® Phase 2 Clinical Trial for HER2 Breast Cancer.
The Company is pleased to confirm that its first Investigational New Drug (IND) application has been submitted to the U.S. Food and Drug Administration (FDA) for formal review and approval, marking a major milestone for the Company. Upon approval of the IND application by the FDA, the Company will commence its Phase 2 Clinical Trial with its strategic trial partner hospitals across the USA. Based on standard FDA review timelines, initiation of the study at multiple sites in the US may begin as early as Q1 2026.
“I am thrilled to announce this milestone to the market and am especially proud of our entire team for delivering a quality submission we can feel confident submitting to the FDA,” said Imagion Biosystems, Inc. President Ward Detwiler. “This is a crucial step in our mission to enable first-class molecular imaging and address a major unmet need in cancer detection. While awaiting response from the FDA, our team has turned its focus to the systems and strategies for expedient and successful completion of this trial.”
MagSense® HER2 Imaging Agent IND Submitted
The IND application marks a major milestone in the clinical development of the MagSense® HER2 imaging agent and the broader MagSense® platform in general: if successful, it indicates that the company has comprehensively addressed all necessary logistical, analytical, manufacturing and quality and safety pre-requisites necessary to conduct the trial. Following submission, the U.S. FDA has 30 days to respond to the company with any questions or comments. The company anticipates opening the study for enrollment following FDA approval.
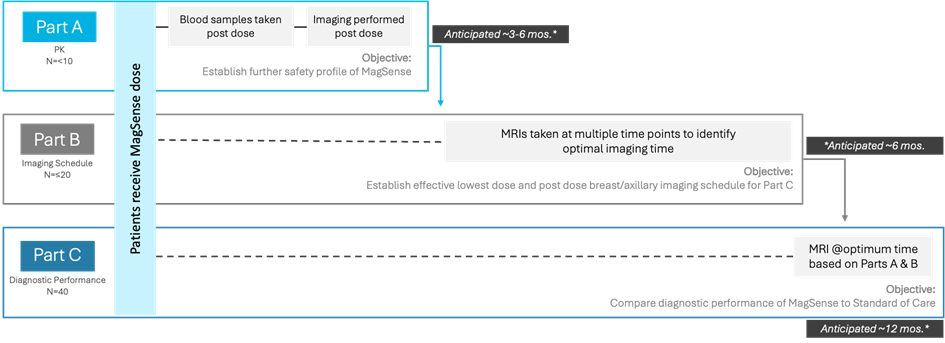
Designed in three parts, the study will start with an initial cohort of subjects to collect additional safety data (Part A), as requested by the US Food and Drug Administration (FDA). The reduced dosing regimen and optimised imaging protocol will then be evaluated in a second group of subjects (Part B) before proceeding to a larger cohort of subjects to establish diagnostic performance (Part C). The MagSense® Phase 2 Clinical Trial for HER2 Breast Cancer is expected to be completed in 18-24 months following acceptance of the IND by the FDA. In addition to validating the diagnostic performance of MagSense® for HER2+ breast cancer, the results of the Phase 2 study will provide valuable insight into the potential impact on cost of care, patient outcomes, and overall clinical value. Additionally, by integrating quantitative imaging techniques into the study protocol, the Phase 2 will yield critical data for the development and training of AI diagnostic tools.
Phase 2 Clinical Trial Design
Strategic Trial Partners Engagement Process Complete
To ensure the planned Phase 2 study can initiate as quickly as possible following acceptance of the IND application by the US FDA, the Company is pleased to confirm that it has selected and engaged all the strategic trial partners required to manage the study. With the IND submitted, these partners have begun the process of clinical-study site engagement and contracting, developing support materials for clinical investigators to expedite participant recruitment, and progressing the logistic and data analysis methods to ensure full compliance with Good Clinical Practices. These activities will help ensure the Company is in a strong position to commence its Phase 2 Clinical Trial launch in early 2026, and well prepared to execute the study successfully and within the prescribed timeframe.

